SUPERIOR HIV PROTECTION VS TRUVADA IN HPTN 083
Fewer HIV-1 acquisitions
- Primary endpoint: 69% reduction in the risk of acquiring HIV-1 with APRETUDE (0.37/100 person-years) vs TRUVADA (1.22/100 person-years) (HR [95% CI]: 0.31 [0.16-0.58]; P=0.0003)
- At the primary analysis, 99.5% of people in the APRETUDE arm and 98.3% of people in the TRUVADA arm remained negative
CI=confidence interval; HR=hazard ratio.
THE APRETUDE CLINICAL TRIALS WERE STOPPED EARLY DUE TO HIGH EFFICACY VS TRUVADA1,5,6
- The independent Data and Safety Monitoring Board recommended the study sponsor stop the blinded phase of the trials and share the results6,7
- According to protocol amendments, study participants were given a choice between either open-label APRETUDE or TRUVADA following the unblinded phase7,8

CONSISTENT PROTECTION ACROSS DIVERSE PATIENT GROUPS IN HPTN 083
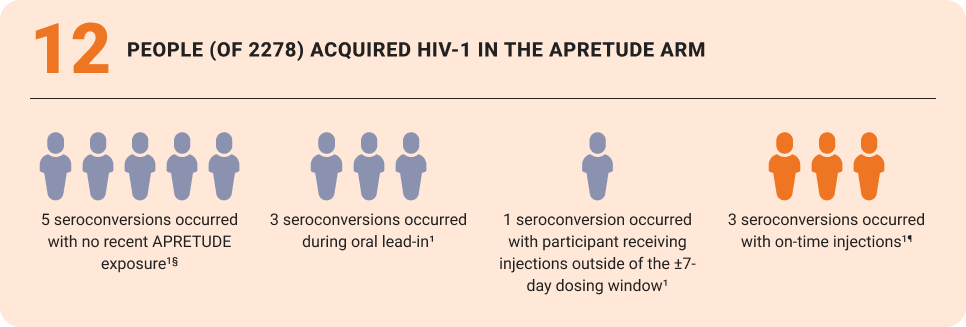
RESISTANCE DATA
- Of the prevalent infections, 1 INSTI-associated mutation was detected in the APRETUDE arm and 2 NRTI-associated mutations were detected in the TRUVADA arm10
#R263K (n=1), E138A+Q148R (n=1), G140A+Q148R (n=1), L74I+E138E/K+G140G/S+Q148R+E157Q (n=1).
||M184l (n=1), M184V (n=2), K65R (n=1).
INSTI=integrase strand transfer inhibitor; NRTI=nucleoside/nucleotide reverse transcriptase inhibitor; RAM=resistance-associated mutation.
PMUS-CBTWCNT250019
References:
Landovitz RJ, Donnell D, Clement ME, et al. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385(7):595-608. doi:10.1056/NEJMoa2101016
Clement ME, Zhe W, Fichtenbaum C, et al. Pre-exposure prophylaxis product choice in US participants in HPTN 083. Poster Presented at: Conference on Retroviruses and Opportunistic Infections; February 19-22, 2023; Seattle, WA. Accessed March 1, 2024. https://www.croiconference.org/abstract/pre-exposure-prophylaxis-product-choice-in-us-participants-in-hptn-083
Delany-Moretlwe S, Hanscom B, Angira F, et al. Initial PrEP product choice: results from the HPTN 084 open-label extension. Abstract presented at: lAS 2023; July 26, 2023; Brisbane, Australia. Accessed March 1, 2024. https://www.hptn.org/sites/default/files/inline-files/220725%20IAS%202023%20product%20choice%20revised.pdf
Safety and efficacy study of injectable cabotegravir compared to daily oral tenofovir disoproxil fumarate/emtricitabine (TDF/FTC), for pre-exposure prophylaxis in HIV-uninfected cisgender men and transgender women who have sex with men. Clinical Trials.gov. November 2023. Accessed January 26, 2024. https://clinicaltrials.gov/study/NCT02720094
Delany-Moretlwe S, Hughes JP, Bock P, et al. Cabotegravir for the prevention of HIV-1 in women: results from HPTN 084, a phase 3, randomised clinical trial. Lancet. 2022;399(10337):1779-1789. doi:10.1016/S0140-6736(22)00538-4
HPTN 084 Study Demonstrates Superiority of CAB LA to Oral TDF/FTC for the Prevention of HIV in cisgender women in sub-Saharan Africa. HIV Prevention Trials Network. Accessed January 26, 2024. https://www.hptn.org/sites/default/files/inline-files/HPTN%20084%20DSMB%20FAQ_V1.0_8Nov2020_0.pdf
Global HIV prevention study to stop early after ViiV Healthcare’s long-acting injectable formulation of cabotegravir dosed every two months shows higher efficacy than daily oral prep. News release. ViiV; May 18, 2020. Accessed October 12, 2023. https://viivhealthcare.com/en-us/media-center/news/press-releases/2020/may/global-hiv-prevention-study-to-stop-early-after-viiv-healthcares
HIV Prevention Trials Network. HPTN 084: A Phase 3 Double Blind Safety and Efficacy Study of Long-Acting Injectable Cabotegravir Compared to Daily Oral TDF/FTC for Pre-Exposure Prophylaxis in HIV-Uninfected Women (protocol version 5.0; October 6, 2023). Accessed February 29, 2024. https://www.hptn.org/research/studies/hptn084
Data on file, ViiV Healthcare.
Marzinke MA, Grinsztejn B, Fogel JM, et al. Characterization of human immunodeficiency virus (HIV) infection in cisgender men and transgender women who have sex with men receiving injectable cabotegravir for HIV prevention: HPTN 083. J Infect Dis. 2021;224(9):1581-1592. doi:10.1093/infdis/jiab152
Eshleman S, Fogel JM, Halvas EK, et al. CAB-LA PrEP: Early detection of HIV infection may reduce INSTI resistance risk. Abstract presented at: Conference on Retroviruses and Opportunistic Infections; February 12-16, 2022; Virtual. Accessed March 1, 2024. https://www.croiconference.org/abstract/cab-la-prep-early-detection-of-hiv-infection-may-reduce-insti-resistance-risk